IDYA Stock Risk & Deep Value Analysis
IDEAYA Biosciences Inc
Healthcare • Biotechnology
DVR Score
out of 10
What You Need to Know About IDYA Stock
We analyzed IDEAYA Biosciences Inc using our deep value framework. Sign in to see our full verdict and DVR Score.
We ran IDYA through our deep value framework — analyzing financial health, distress signals, competitive moat, and risk factors. Our risk assessment: Aggressive. Here's what we found.
How Risky Is IDYA Stock?
Overall Risk
Aggressive
Financial Risk
Medium
Market Risk
High
Competitive Risk
Medium
Execution Risk
Medium
Regulatory Risk
High
What Are the Red Flags for IDYA?
- ⚠
Negative or inconclusive clinical trial results for Darovasertib or IDE161
- ⚠
Regulatory delays or setbacks from FDA/EMA
- ⚠
Significant safety concerns emerging from ongoing trials
- ⚠
Increased competitive pressure from other companies developing synthetic lethality targets or alternative oncology treatments
Unlock IDYA Red Flags & Risk Warnings
Create a free account to see the full analysis
What Does IDEAYA Biosciences Inc (IDYA) Do?
Market Cap
$3.13B
Sector
Healthcare
Industry
Biotechnology
Employees
131
IDEAYA Biosciences, Inc., a precision medicine oncology company, discovers and develops targeted therapeutics for patient populations selected using molecular diagnostics in the United States. The company's products in development include IDE196, a protein kinase C inhibitor that is in Phase 2/3 clinical trials for genetically defined cancers having GNAQ or GNA11 gene mutations; IDE397, a methionine adenosyltransferase 2a inhibitor that is in Phase 2 clinical trials for patients with solid tumors having methylthioadenosine phosphorylase gene deletions, such as non-small cell lung (SCLC) and MTAP-deletion urothelial cancer; IDE849, a DLL3 antibody drug conjugate inhibitor that is in Phase 1 clinical trial for the treatment of SCLC and neuroendocrine tumors; IDE275, a Werner Helicase inhibitor that is in Phase 1 clinical trial for patients having tumors with high microsatellite instability; IDE161, a poly ADP-ribose glycohydrolase inhibitor that is in Phase 1/2 clinical trial to treat tumors with homologous recombination deficiency (HRD), and other genetic or molecular signatures; IDE705 (GSK101), a Poly Theta Helicase inhibitor that is in Phase 1 clinical trial for the treatment of tumors with BRCA or other mutations associated with HRD. It also develops preclinical programs, including IDE892, a MTA-cooperative PRMT5 inhibitor; IDE034, a B7H3/PTK7 topo-I-payload bispecific BsADC; and IDE251, a KAT6/7 inhibitor. In addition, the company offers preclinical research programs focused on pharmacological inhibition; DECIPHER dual CRISPR library for synthetic lethality target and biomarker discovery; and INQUIRE chemical library and HARMONY machine-learning engines to enhance its drug discovery platform. The company has strategic alliances with GlaxoSmithKline, Pfizer Inc., Gilead Sciences, Inc., Merck, Biocytogen, Cancer Research UK, and the University of Manchester. IDEAYA Biosciences, Inc. was incorporated in 2015 and is headquartered in South San Francisco, California.
Visit IDEAYA Biosciences Inc WebsiteIs IDYA Stock Undervalued?
Unlock the full AI analysis for IDYA
Get the complete DVR score, risk analysis, and more
Does IDYA Have a Competitive Moat?
Sign in to unlockMoat Rating
🛡️ Narrow
Moat Trend
Expanding
Moat Sources
3 Identified
The moat is primarily built on the company's proprietary synthetic lethality platform and the intellectual property protecting its drug candidates. As more programs advance through clinical trials and potentially gain regulatory approval, the moat strengthens through data exclusivity and market leadership in specific novel targets. The GSK partnership further validates and expands this moat through shared expertise and resources.
Moat Erosion Risks
- •Clinical trial failures, invalidating the efficacy or safety of their lead assets
- •Expiration or invalidation of key patents, allowing competitors to develop biosimilars or similar drugs
- •Emergence of superior competing technologies or treatment paradigms
IDYA Competitive Moat Analysis
Sign up to see competitive advantages
What Could Drive IDYA Stock Higher?
Near-Term (0-6 months)
- •Q1 2026 Earnings Report (Estimated early May 2026)
- •Presentation of updated clinical data for Darovasertib (uveal melanoma) at major oncology conferences (Q2/Q3 2026)
- •Initial Phase 1/2 clinical data readout for IDE161 (HRD tumors) in Q2 2026
Medium-Term (6-18 months)
- •Progression of IDEAYA's other synthetic lethality programs (e.g., Pol Theta, PARP/PARG inhibitors) into clinical trials (late 2026 / early 2027)
- •Potential expansion of GSK partnership or new strategic collaborations (2027)
- •Additional milestone payments from GSK as programs advance
Long-Term (18+ months)
- •Regulatory filings and potential approval for Darovasertib in uveal melanoma (2028-2029)
- •Establishment as a leading synthetic lethality platform company, with multiple approved drugs (2030+)
- •Industry-wide shift towards precision oncology and targeted therapies
Catalysts & Growth Drivers
Upgrade to Premium to see catalysts
What's the Bull Case for IDYA?
- ✓
Positive clinical trial data (efficacy and safety) for Darovasertib and IDE161
- ✓
Successful progression of other synthetic lethality programs into the clinic
- ✓
Achievement of additional milestone payments from GSK
- ✓
Maintenance of a strong cash runway and efficient capital allocation
Bull Case Analysis
See what could go right with Premium
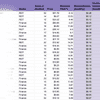
Compare IDYA to Similar Stocks
See how IDEAYA Biosciences Inc stacks up against related companies in our head-to-head analysis.
📊 Explore More Stock Analysis
Get comprehensive Deep Value Reports for thousands of stocks. Research risk, financial health, and investment potential with our AI-powered analysis.
Important Disclaimer – Not Financial Advice
Deep Value Reports is an independent research platform for educational and informational purposes only. We are not financial advisors, investment advisors, or licensed professionals. The analysis, scores, and information provided on this page for IDYA (IDEAYA Biosciences Inc) should not be construed as personalized investment advice, a recommendation to buy or sell any security, or an offer to provide investment advisory services.
All investments involve risk, including the potential loss of principal. Past performance does not guarantee future results. Always conduct your own research, consider your financial situation, and consult with a qualified financial advisor before making any investment decisions.